Table of contents
Lithium-ion batteries, composed of various individual cells, are particularly powerful due to their structure and the materials used. The robust construction of lithium-ion batteries guarantees high performance and durability over long periods of operation. Intermediate charges are possible at any time with these innovative rechargeable batteries, and they can be used in multi-shift operation without any issues. Find out more about lithium-ion battery manufacturing including which metals they contain and how the batteries work in our professional guide.
Small power pack: components of lithium-ion batteries
A lithium-ion battery is composed of many individual cells. Each of these cells always has the same structure and contains the following components:
- Positive electrode: The cathode of the lithium-ion battery consists of lithium metal oxide, which may contain variable amounts of nickel, manganese and cobalt. The metal oxides are also called transition metal.
- Negative electrode: The anode is usually made of graphite.
- Electrolyte: In order for the lithium ions to move as charge carriers in the cell, anhydrous electrolyte can also be found in the battery. This contains salts such as lithium hexafluorophosphate dissolved in an aprotic solvent such as diethyl carbonate. In lithium polymer batteries, a polymer of polyvinylidene fluoride or polyvinylidene fluoride hexafluoropropylene is used at this point.
- Separator: To avoid short circuits, a separator made of non-woven materials or polymer films is installed between the electrodes. The separator is permeable to lithium ions and can absorb large quantities of the ions.
The separator — essential for the safe functioning of lithium-ion batteries
The separator built into the lithium-ion batteries controls and secures the electrochemical reactions inside the lithium-ion battery. On the one hand, it isolates the two electrodes from each other so that internal short circuits cannot occur. At the same time, the special permeable design ensures that only lithium ions can pass through and thus move between the negative and positive electrodes. In addition, the separator ensures gas exchange in the closed lithium battery cells.
For this purpose, the component must be made of microporous membranes, which can differ depending on the battery power and size. Either polymeric films (as in lithium-polymer batteries) or heat-resistant ceramic separators are used for this purpose. By combining non-wovens with a ceramic coating during lithium-ion battery construction, separators are particularly flexible and yet temperature-resistant up to 700°C.
The battery management system — optimising the functions
When multiple cells are combined into a module, it typically includes an integrated control system. The most important electrical component in the construction of a lithium-ion battery is the battery management system (BMS). This consists of several parts: the cell supervision unit (CSU), the battery control unit (BCU), and the battery disconnect unit (BDU). As a whole, the BMS acts as an interface between the device and the battery. It also optimises the capacity, energy, and performance of the accumulator. It also prevents deep discharge even when the lithium-ion battery is in storage for a longer period of time, thus extending the service life.
High-quality metals for a functional construction of Li-ion batteries
The design, which allows lithium to move back and forth between the electrodes in ionised form, also gives the name to lithium-ion batteries. Depending on the electrode materials used, Li-ion batteries are divided into different groups. The mode of operation remains the same, but the energy density, cell voltage, temperature sensitivity, capacity, and permitted charge and discharge current can vary due to the use of different transition metal ions. The construction of lithium-ion batteries is as follows:
- Lithium-polymer batteries: The electrolyte used here is a polymer-based film with a gel-like consistency. This structure makes it possible to produce particularly small batteries (less than 0.1 mm thick) in various designs. With an energy density of up to 255 Wh/kg, they are very powerful, but mechanically, electrically and thermally sensitive.
- Lithium cobalt dioxide batteries: The positive electrode of this type of battery consists of lithium cobalt dioxide. The anode is made of graphite. These types of batteries are prone to thermal runaway when overloaded.
- Lithium titanate batteries: Here, the negative electrodes are not made of graphite, but of sintered lithium titan spinel. These enable an outstanding fast-charging capacity as well as operation at temperatures as low as -40° Celsius. The positive electrodes, in turn, consist of lithium titanium oxide..
- Lithium iron phosphate batteries: Cells of this battery type each have a cathode made of lithium iron phosphate. The electrolyte is present here in solid form. These types of batteries have a lower energy density of up to 110 Wh/kg but are not prone to thermal runaway if mechanically damaged. The discharge voltage curve indicates a memory effect in these batteries, but this is very low compared to NiCd batteries.
Good to know: the raw materials used in these batteries are not only valuable but also potentially harmful to the environment. As a result, there are specific regulations governing the disposal of lithium-ion batteries. These rules ensure proper recycling of components and prevent environmental damage. Additionally, because lithium batteries are classified as dangerous goods, special regulations apply to their transport as well.
How lithium-ion batteries function during charging and discharging
Lithium-ion batteries function according to a simple principle: the electrical energy in the lithium-ion batteries is stored through a chemical process and made usable for the propulsion of pick-up devices such as electric stacker trucks.
The mode of operation is essentially based on the constant movement of ionised lithium between the electrodes. The lithium-ion flow balances the external current flow when charging and discharging the batteries, so that the electrodes themselves remain electrically neutral:
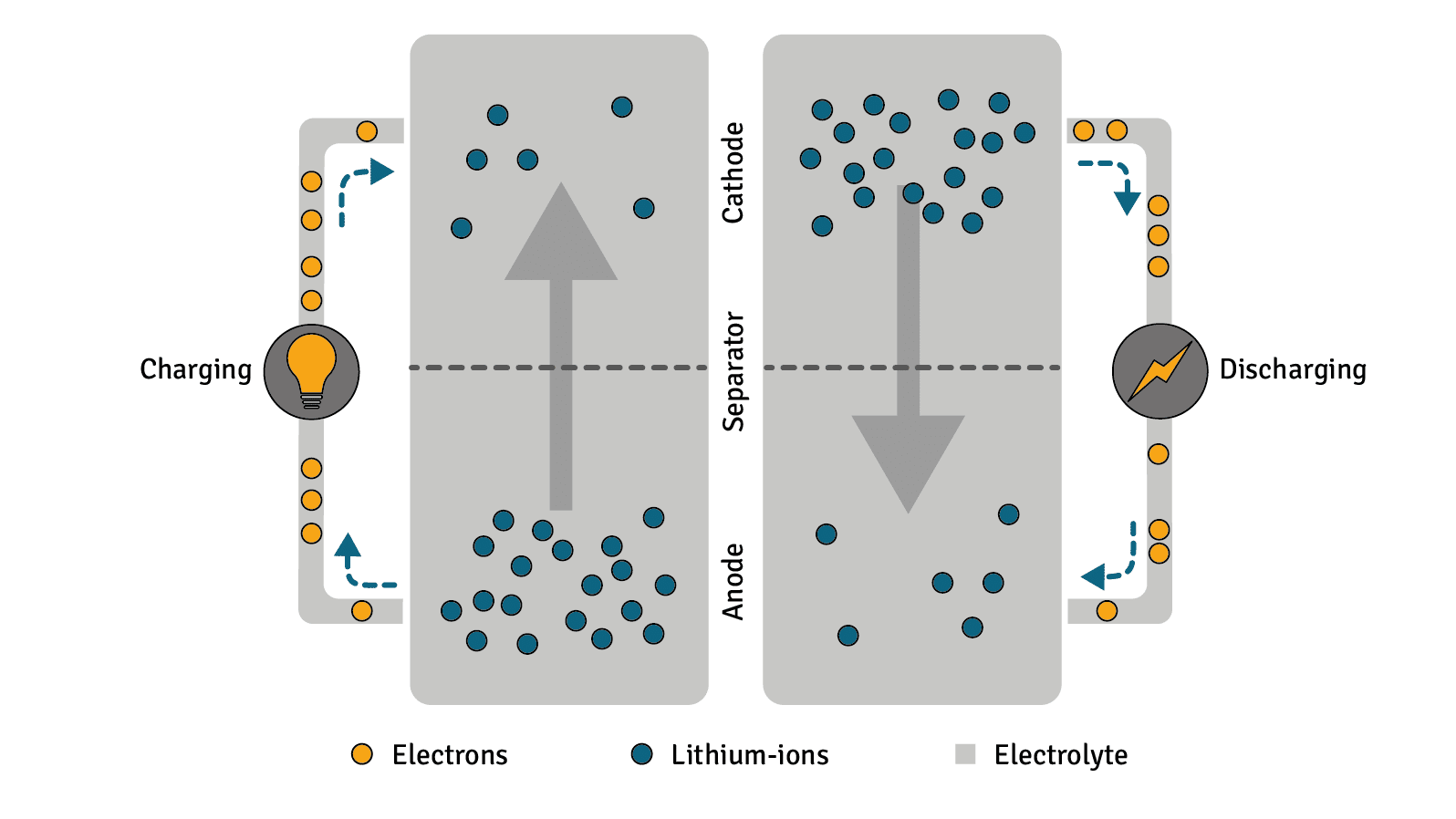
1. Discharge
If the battery is discharged – i.e., its stored energy is used by a terminal device – the lithium atoms each emit an electron at the negative electrode. This electron is returned to the positive electrode via the external circuit. In the same step, the same number of lithium ions move from the negative electrode through the electrolyte and the separator to the positive electrode. The electrons are picked up at the positive electrode by strongly ionised transition metal ions. These can be different depending on the type of battery. Unlike lithium ions, they are not mobile.
2. Charge/Recharge
When charging the accumulator cells, the non-ionised lithium atoms move from the positive electrode through the separator back to the negative electrode. Here they are intercalated between graphite molecules. This process, also called intercalation, is triggered by charging with constant current until the nominal current is reached. When the end-of-charge voltage is reached, it is maintained while the charging current decreases.
FAQ on lithium-ion batteries construction and function
Lithium-ion batteries function according to a simple principle: electrical energy in the lithium-ion batteries is stored by a chemical process and made usable for the drive of pick-up devices. The mode of operation is essentially based on the constant movement of ionised lithium between the electrodes. The lithium-ion flow balances the external current flow when charging and discharging the batteries, so that the electrodes themselves remain electrically neutral
The construction of the lithium-ion battery management system fulfils different functions. It controls the charging and discharging of the Li-ion battery and acts as an interface between the device and the battery. In addition, the BMS optimises the capacity, energy and performance of the battery, prevents deep discharge and thus extends the service life.
The separator built into lithium-ion batteries controls and secures the electrochemical reactions inside the lithium-ion battery. On the one hand, it isolates the two electrodes from each other so that internal short circuits cannot occur. At the same time, the special permeable design ensures that only lithium ions pass through and thus move between the negative and positive electrodes. In addition, the separator ensures gas exchange in the closed lithium battery cells. The separator therefore fulfils an important safety function.
Li-ion batteries can contain different metals, such as lithium, nickel, cobalt, manganese, aluminium, titanium or copper.
Image source:
© gettyimages.de – alengo









